Imagine testing tap water for hidden dangers or crafting dyes that won’t fade after years. That’s where dimethyl-p-phenylenediamine, often called DMPD, steps in. This simple chemical plays a big role in keeping our water clean and our products durable. It acts as a key player in tests and manufacturing processes we rely on every day.
In this guide, we’ll explore DMPD’s basic makeup, its top uses in water checks and industry, and how to handle it safely. You’ll get clear facts on its structure, real-world applications, and tips to avoid risks. Whether you’re a lab tech or just curious, this guide covers what you need to know about dimethyl-p-phenylenediamine.
Chemical Foundation: Understanding Dimethyl-p-Phenylenediamine (DMPD)
DMPD begins its journey as a fundamental component in chemistry laboratories and manufacturing facilities. Its formula, C₈H₁₂N₂, shows two nitrogen atoms linked to a benzene ring with methyl groups attached. Think of it like a modified version of p-phenylenediamine, where those extra methyl bits change how it reacts.
Section 1: Molecular Structure and Synthesis
N,N-Dimethyl-p-phenylenediamine derives its name based on the positions of these groups relative to each other on the ring. The “p” stands for para, which is opposite. You synthesize it by adding methyl groups to p-phenylenediamine through a reaction with formaldehyde and an acid or by reducing other compounds. This molecule is a simple compound to modify for different purposes. In labs, chemists control the steps needed to get pure DMPD. Its structure lets it donate electrons fast, which is why it shines in tests.
Physical and Chemical Properties
DMPD appears as white to light brown crystals or powder. It melts at about 54°C and boils higher, around 262°C. You can dissolve it well in water, alcohol, or ether, but it hates strong light or air. Oxidation is the main trick. When oxygen hits it, DMPD turns red or magenta. This color shift is gold for spotting chemicals like chlorine. It stays stable in dry form but breaks down in wet solutions over time. Reactivity comes from those amine groups. They bond easily with oxidants. Keep it cool and dark to slow down any changes.
Purity Grades and Common Derivatives
Suppliers offer DMPD in 98% pure form for lab work or lower for bulk industry use. The sulfate salt version dissolves better and stores longer. Analytical grades hit 99% purity to avoid errors in tests. Derivatives include salts like hydrochloride for specific reactions. Choose based on your job—high purity for precise measurements, standard for making dyes. Always inspect labels for contaminants.
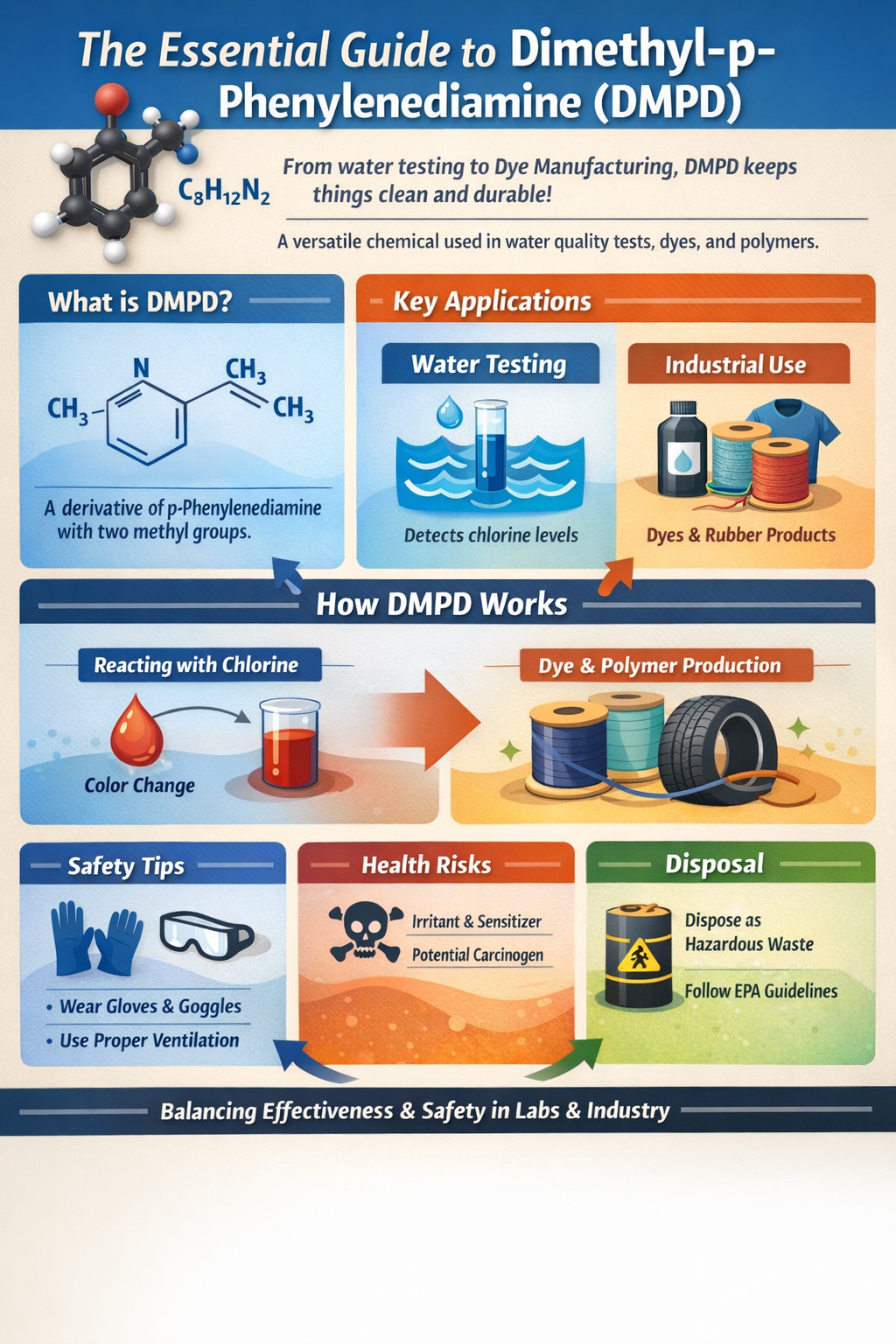
Section 2: Primary Application: DMPD in Water Quality Testing
Water safety matters to everyone. DMPD helps detect chlorine, the germ-killer in pools and taps. Without it, we might miss low levels that affect health.
The Chemistry of Colorimetric Analysis
In the DPD method, DMPD reacts with free chlorine to form a red color. Add a buffer and the chemical, then measure the hue with a device or by eye. The deeper the red, the more chlorine presents. This works because chlorine oxidizes DMPD, pulling electrons and creating Wurster’s dye. It’s quick results in seconds. Labs use it to ensure water stays disinfected without excess chemicals that harm pipes or taste. You see this process in municipal plants daily. One drop of the DMPD solution can flag issues fast, saving time over slow lab sends.
Comparison with Other Chlorine Testing Methods
The DPD method beats iodometric titration for field work. Titration needs extra tools and takes minutes, while DPD provides instant reads. It’s more sensitive too, spotting 0.02 mg/L of chlorine. Compared to test strips, DPD handles samples better without interference from metals. Strips fade quick; DPD kits last longer. For on-site checks, like at beaches or spas, DPD wins with ease. Drawbacks? It reacts only with free chlorine, so total checks need tweaks. Still, pros pick it for reliability.
Standardization and Regulatory Acceptance
EPA method 4500-Cl G endorses the DPD test for drinking water. It’s standard in many countries too. The WHO nods to it for global monitoring. Regulations demand accurate readings to track byproducts like trihalomethanes. DMPD helps meet those rules. In 2025 data, over 80% of U.S. utilities used DPD-based kits for compliance. Stick to certified reagents to match these standards.
Section 3: Industrial Roles: DMPD in Polymer and Dye Manufacturing
Beyond water, DMPD builds tough materials. It steps into factories where color and strength count.
Function as a Chemical Intermediate
DMPD serves as a starter for azo dyes, bright colors in fabrics and inks. It couples with other chemicals to lock in shades that resist washing. Think of it as the glue for vibrant blues or reds. In dyeing plants, it cuts costs by reacting efficiently. One batch yields a ton of product. Variations, like long tail use in eco-dyes, show their flexibility.
Utilization in Rubber and Polymer Chemistry
Add DMPD to rubber mixes as an antiozonant. Ozone cracks tires; this chemical blocks it by scavenging radicals. Synthetic rubbers in cars or belts last years longer. It also acts as an antioxidant, stopping heat from breaking chains. Factories blend it at 1-2% levels. Results? Products that flex without crumbling.
Photographic and Imaging Applications (Historical Context)
Back in the film era, DMPD developed black-and-white photos. Its redox power pulled silver from emulsions. Though digital took over, niche labs still use it for specialty prints. Today, it pops up in old-school processes or research. The color change mimics early color films. It’s a nod to chemistry’s past.
Section 4: Safety, Handling, and Environmental Considerations
DMPD isn’t all fun. It can irritate skin or worse if mishandled. Know the risks to stay safe.
Occupational Exposure Limits and Health Hazards
OSHA sets a limit of 0.1 mg/m³ for air exposure. Skin contact causes rashes; it’s a sensitizer. Long-term animal studies link it to cancer risks, so treat it as a possible carcinogen. Eyes sting on contact; inhaling fumes leads to headaches. SDS sheets warn of these. Wear protection every time. Inhaled dust affects lungs. Gloves prevent absorption through skin.
Best Practices for Storage and Handling
Store DMPD in cool, dark spots below 25°C. Use glass or plastic bottles sealed tight. Air turns it brown, so a nitrogen flush helps. When weighing, do it under a hood with goggles and nitrile gloves. Avoid cotton—it soaks up spills. Clean tools with water right away.
For spills, absorb them with sand and rinse the area thoroughly. Train teams on these steps.
- Wear full PPE: gloves, goggles, and a lab coat.
- Work in ventilated areas.
- Label all containers clearly.
Environmental Fate and Disposal Protocols
DMPD breaks down in soil but sticks in water, harming fish. It oxidizes to toxic dyes. Don’t dump in drains—it clogs treatment plants. Neutralize waste with bleach before disposal. Send to chemical waste services. EPA rules ban direct release. Follow local guidelines. Recycle pure scraps if possible.
Conclusion: The Indispensable Role of Dimethyl-p-Phenylenediamine
Dimethyl-p-phenylenediamine, or DMPD, serves as the crucial link between laboratory testing and factory floors. DMPD operates discreetly, whether it’s aiding in the detection of chlorine in your glass or prolonging the lifespan of your shoes’ rubber. Its simplicity conceals its immense power. If you understand the chemistry behind DMPD, you can make the most out of it. It’s a balancing act between benefits and safety, which can make all the difference. Quality control measures in 2026 will only make DMPD more relevant. The next time you test water or apply a color that won’t fade, remember this unheralded worker. It’s up to you to make the most out of it. For more information about chemicals like DMPD, try searching online sites that provide lab supplies and safety information.

Leave A Comment