Consider this scenario. Upon opening the bottle of diethyl ether in the laboratory setting, it takes just seconds for the room to be filled with a strong odor, which appears sweet at the same time. The diethyl ether almost seems to disappear right before your eyes, leaving you with a completely empty bottle and a foggy workspace. Extremely rapid evaporation from the liquid phase to the gaseous one significantly contributes to the extensive use of diethyl ether in medicine and chemistry studies.
Which mechanisms make it happen? The molecular structure of the compound, along with its weak intermolecular connections, is the primary reason for the compound’s high volatility. Thus, the low boiling point and high vapor pressure contribute to a rapid transformation of the liquid to the gaseous state under normal conditions.
Defining Volatility and Vapor Pressure in Solvents
1. What Makes a Compound Volatile?
Volatility means how fast a liquid turns into vapor. For diethyl ether, this happens quickly because its molecules slip away from the liquid state with ease. Think of it like water versus gasoline—water sits around longer, while gasoline vanishes fast on a hot day. Highly volatile compounds, such as diethyl ether, have low boiling points. This lets them change phases without much heat. Less volatile ones, like glycerol in hand sanitizer, need more energy to evaporate. The link between boiling point and volatility is simple: lower boiling point equals faster evaporation. You see this in everyday life. A drop of ether on your skin feels cold right away as it evaporates, pulling heat from your body. That chill shows how eager its molecules are to escape.
2. The Key Role of Vapor Pressure
Vapor pressure measures the push from gas molecules above a liquid. In diethyl ether, this pressure stays high at room temperature, around 442 mmHg at 20°C. That means many molecules gain enough speed to break free and join the air. Compare that to acetone at about 185 mmHg or water at just 17 mmHg under the same conditions. Ether’s high vapor pressure drives its rapid spread into the atmosphere. It’s why you smell it instantly—those vapor molecules rush out. This trait makes diethyl ether a top pick for quick-drying tasks. But it also means you must handle it with care to avoid building too much vapor in closed spaces.
3. Boiling Point as a Volatility Gauge
Diethyl ether boils at 34.6°C, just above room temperature. That’s why it feels so fleeting; a small warmth spike sends it into vapor form. At body temp, around 37°C, it’s already past that point, which adds to its tricky nature in medical settings. This low boiling point ties right into why diethyl ether is highly volatile. It needs little energy to shift phases, unlike water’s 100°C hurdle. In labs, this makes ether easy to remove after use, but it demands watchful eyes.
Data backs this up: Ether’s boiling point sits way below many solvents, like benzene at 80°C. That gap explains the speed difference in evaporation rates.
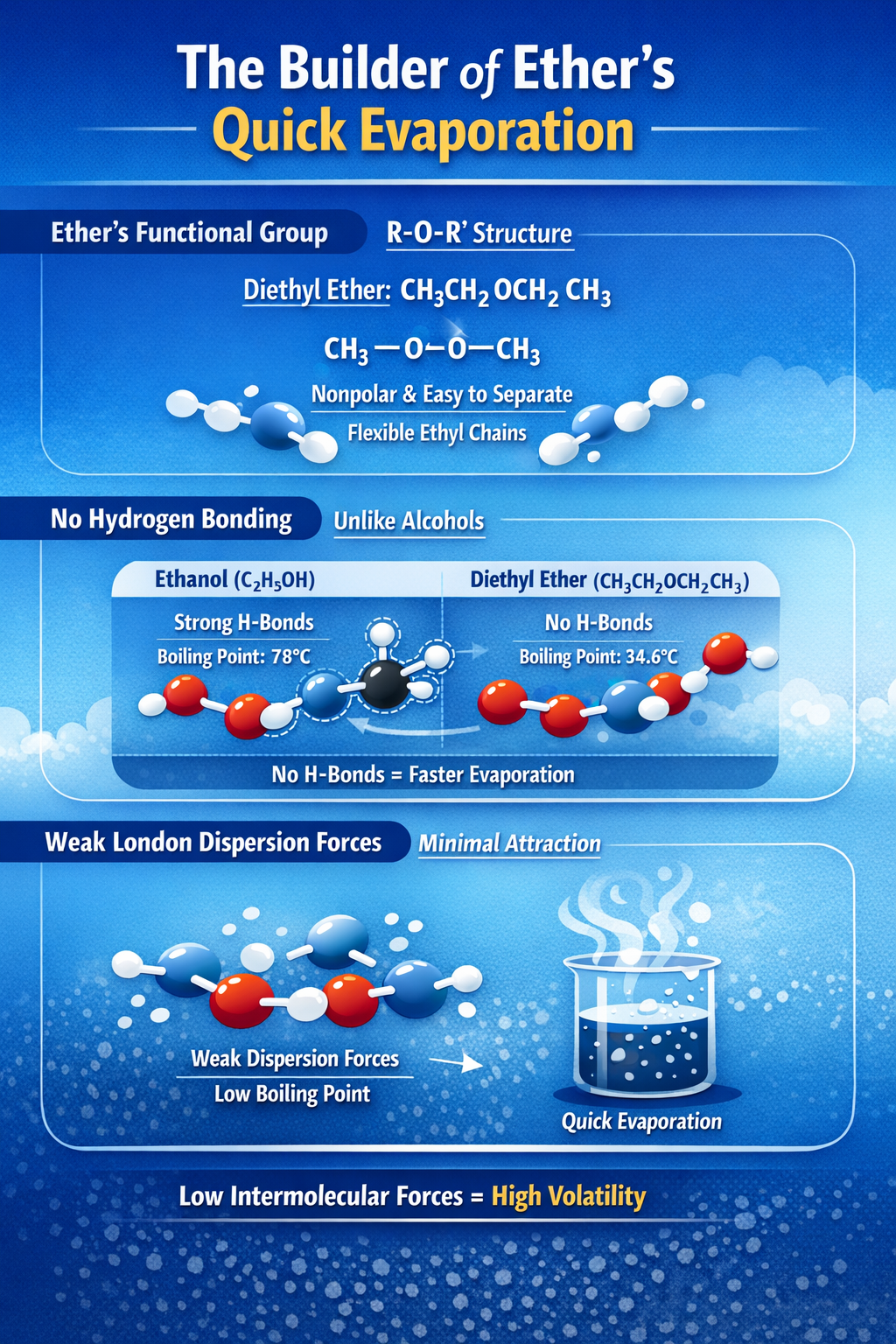
Molecular Structure: The Builder of Ether’s Quick Evaporation
i. Ether’s Special Functional Group (R-O-R’)
Diethyl ether’s formula is CH₃CH₂OCH₂CH₃, with an oxygen atom linking two ethyl chains. This ether group, R-O-R, gives it a mostly nonpolar vibe, even with oxygen in the mix. The oxygen pulls electrons but doesn’t make the whole molecule polar like in water. That structure keeps things light and non-sticky. The ethyl groups are like flexible arms, letting molecules slide past each other. No heavy polar pull means less hold on the liquid form. You can picture it as oil on water—ether doesn’t cling tight. This setup fuels its high volatility by easing molecule separation.
ii. No Hydrogen Bonding, Unlike Alcohols
Here’s the big split: Diethyl ether lacks the O-H bond found in alcohols like ethanol. Ethanol boils at 78°C because hydrogen bonds create strong links between molecules. Ether? It skips those bonds, so attractions stay weak. Without hydrogen bonding, ether molecules don’t team up firmly. Ethanol’s extra stickiness raises its boiling point by over 40°C. That’s why ether evaporates so much faster—fewer barriers to overcome.
Compare the two side by side:
- Ethanol: Strong H-bonds, slower vapor loss.
- Diethyl ether: No H-bonds, quick phase shift.
This absence is why chemists favor ether for tasks needing rapid drying.
iii. Weak London Dispersion Forces at Play
London dispersion forces are the main glue for diethyl ether molecules. These temporary attractions arise from electron wiggles, but they’re faint compared to hydrogen bonds. In ether, the nonpolar chains make these forces even weaker. The weak links are easily broken, resulting in the low boiling point we observe. At room temperature, most molecules have enough jiggles to pull them away. Water’s stronger forces keep it liquid longer. Ether’s chain length adds a touch of dispersion, but not enough to slow things down much. This concentration is the ideal level of volatility without reaching extreme levels.
Kinetic Energy and the Drive to Evaporate
i. Kinetic Energy Spread at Room Temp.
Molecules buzz with kinetic energy, distributed along a curve called the Maxwell–Boltzmann distribution. At 25 °C, more diethyl ether molecules have enough energy to escape the liquid than in water. Ether’s weak intermolecular forces lower the energy barrier, so a larger fraction of its molecules can vaporize right away. That spread of energies explains why ether feels so lively: while most water molecules stay below the threshold for vaporization, ether’s faster-moving molecules rush into the air. It’s like a crowd where many push toward the exit while most of the others stay put. That energy edge makes diethyl ether highly volatile at everyday conditions—no stoves or heaters needed, just air.
ii. How Surface Area Affects Evaporation Speed
The evaporation rate hinges on the exposed surface. A wide spill of ether turns into gas quicker than the same amount in a thin tube. Volatility is the built-in tendency, but area amps the action. Spill a puddle on the floor, and vapors rise fast, filling the room. In a flask with a small opening, it takes longer, but the drive stays the same. Wind or heat can boost this even more.
Real labs use these techniques: Spread ether thinly for quick cleanup or contain it to control the pace. Either way, its core volatility shines through.
Real-World Effects of Ether’s Fast Evaporation
i. Lab Safety: Watching Out for Fire Risks
High volatility spells trouble with flames. Diethyl ether’s vapors ignite easily, with a flash point of -45°C. That rapid evaporation builds flammable clouds near the source, turning a small spill into a hazard. Always work in a fume hood to whisk vapors away. No open flames—sparks from switches count too. Labels scream this: “Extremely flammable—keep away from ignition.” Stats show why: Ether causes more lab fires than slower solvents. Smart handling keeps risks low.
ii. Uses in Industry: Extraction and Reactions
Ether’s quick vanish makes it ideal for pulling compounds from mixes. In solvent extraction, you shake with ether, then let it evaporate to grab the prize. No high temperature is needed, saving time and energy. It’s also a reaction medium where you want the solvent to go quickly after post-processing. Think about drug making or perfume blending—either slips away, leaving pure results. That volatility transforms a difficult task into an effortless one. Industries pick it for green methods too, as low temps cut power use.
iii. Past Role in Medicine: Quick Anesthesia Hits
Back in the 1800s, doctors inhaled ether for surgery. Its high volatility meant rapid uptake into blood via lungs, knocking you out quickly. Once stopped, the vapors cleared quickly, leading to a rapid wake-up. Crawford Long used it first in 1842, and it spread wide. The rapid onset and offset beat slower options like chloroform. But risks from volatility—fire and nausea—pushed better drugs later. In today’s world, we may give a nod to that history while using safer picks. Still, Ether’s traits shaped early care.
Conclusion: Grasping the Ether Effect
Diethyl ether’s high volatility boils down to weak London forces, no hydrogen bonding, and a low 34.6°C boiling point. These mix to create strong vapor pressure, letting it evaporate at everyday temps. From labs to history, this trait drives both perks and pitfalls. Next time you spot that sweet smell and recall the molecule magic at work. Dive deeper into organic chemistry and grab a book or hit a lab demo. Understanding these basics keeps science safe and fun for all.

Leave A Comment